Menu
Rx Generic Product Catalog
Therapeutic Class: Oral Contraceptive
TE Code:
AB
AB
Therapeutic Equivalence [TE] Code:
Products meeting necessary bioequivalence requirements
OTC/Rx:
Rx
Rx
Requires a Prescription
Important Product Resources:
Including Black Box Warning
and Patient Labeling
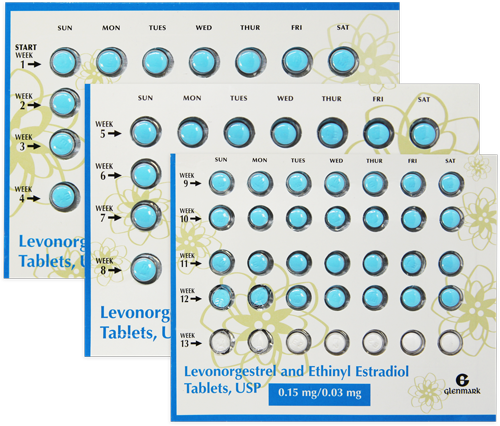
Strength: 0.15 mg/0.03 mg
Description:
(84)
Blue,
Round Tablets [Levonorgestrel and Ethinyl Estradiol]
(7)
White,
Round Tablets [Inert]
Available Sizes [NDC & Pack]:
68462-0672-95
3 x 91 Carton

Imprint Side
1: E1
Imprint Side
2: Plain
Description:
Blue, Round Tablets [Days 1-84]
Blue, Round Tablets [Days 1-84]
Strength: 0.15 mg/0.03 mg
Contains:
Levonorgestrel and Ethinyl Estradiol
Levonorgestrel and Ethinyl Estradiol

Imprint Side
1: E2
Imprint Side
2: Plain
Description:
White, Round Tablets [Days 85-91]
White, Round Tablets [Days 85-91]
Strength: n/a
Contains:
Inert
Inert




